Pourbaix diagram
The traditional method to plot a Pourbaix diagram:[edit]
Here is the link of wikipedia explains what is Pourbaix diagram and how to make it.
https://en.wikipedia.org/wiki/Pourbaix_diagram
I am not going to repeat it here but will show how to make it easier. The general processes can be listed as follows.
- List all possible reactions between all compounds
- Calculate the Gibbs free energy change of each reaction
- According to the number of electrons, Hydrogens to plot vertical lines, horizontal lines and sloped lines.
Usually, there are lots of possible reactions. For a metal with 10 possible phases, the number of possible reaction is <math>C_{10}^2</math>. We can reduce the number according to our experience or some methods. As the solid metal exists at low pH and low potential. We can start from the region, then go higher pH and higher potential. let's take the Ni-H2O as an example.
<math>Ni \leftrightarrow NiO (Ni(OH)2)</math>
<math>Ni \leftrightarrow NiO_2 </math>
<math>Ni \leftrightarrow Ni_3O_4 </math>
<math>Ni \leftrightarrow Ni_2O_3 </math>
<math>Ni \leftrightarrow Ni(OH)_4^{2-} </math>
<math>Ni \leftrightarrow Ni(OH)_3^{-} </math>
<math>Ni \leftrightarrow Ni^{2+} </math>
<math>Ni \leftrightarrow Ni^{2+} </math>
<math>Ni \leftrightarrow NiOOH </math>
If you calculate it, then you will know the boundary of Ni. <math>Ni^{2+}</math>, <math>NiO</math>, and <math>Ni(OH)_4^{2-}</math> are on the top above(higher potential) of the Ni metal phase from low pH to high pH.
Then you can calculate the boundaries of <math>Ni^{2+}</math>, <math>NiO</math>, and <math>Ni(OH)_4^{2-}</math>. If you do it in this sequence, you will skip some reactions involved with meta-stable phases.
A universal reference method[edit]
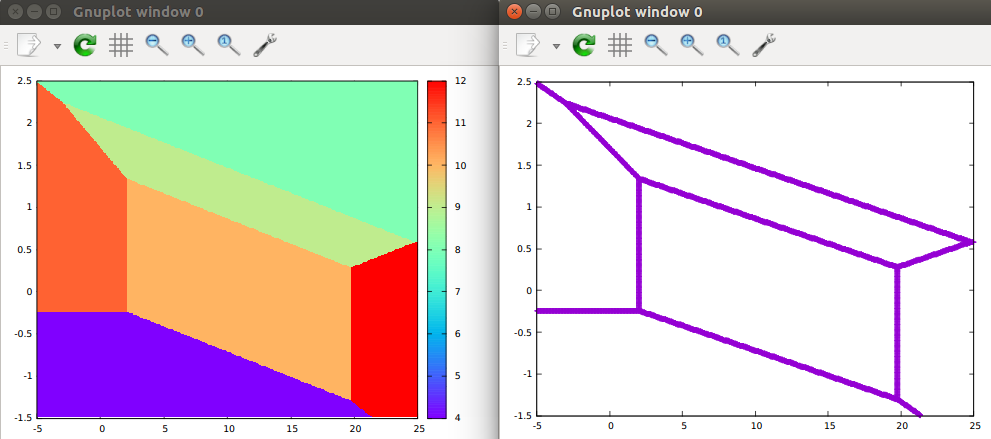
I developed a new method based on Hess' law. I attached the illustration here.[1] In this method, you only need to consider n reactions, where n is the number of phases. I have written a code to deal with this. So, you only need to input the Gibbs free energy of each phase and plot the Pourbaix diagram use gnuplot. Left and right are figures produced by “gnuplot ni.gnulopt” and “gnuplot nil.gnulopt”, respectively.
The code will be available on our group repository.[2]